April 02, 20240 min read
Next-level SxMD efficiency and integrity with Matrix 2.4.1
 Read more
Read more What is a Device History Record (DHR)?
What is a Device History Record (DHR)?April 26, 20240 min read
Read more What is a Quality Audit?
What is a Quality Audit?April 24, 20240 min read
Read more What is the Device Master Record (DMR)?
What is the Device Master Record (DMR)?April 23, 20240 min read
Read more FDA Cleared vs. Approved
FDA Cleared vs. ApprovedApril 19, 20240 min read
Read more IQ, OQ, PQ: Your essential guide to Process Validation
IQ, OQ, PQ: Your essential guide to Process ValidationApril 18, 20240 min read
Read more Item-based vs document-based approach to documentation in Medical Devices
Item-based vs document-based approach to documentation in Medical DevicesApril 17, 20240 min read
Read more 510k Database for Medical Devices
510k Database for Medical DevicesApril 16, 20240 min read
Read more Navigating Innovation: A Comprehensive Guide to the FDA De Novo Pathway for Medical Devices
Navigating Innovation: A Comprehensive Guide to the FDA De Novo Pathway for Medical DevicesApril 15, 20240 min read
Read more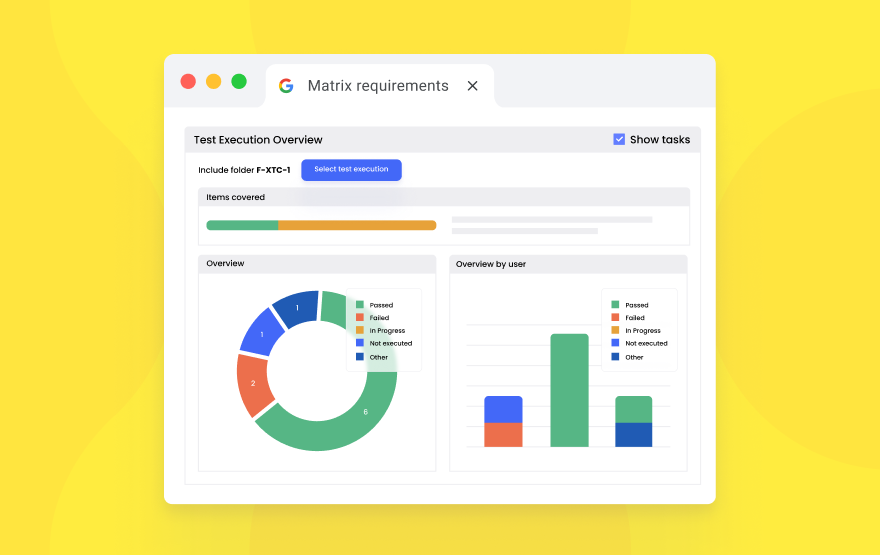 Medical Device Clinical Trials: Everything you need to know about Clinical Testing & Regulatory Pathways.
Medical Device Clinical Trials: Everything you need to know about Clinical Testing & Regulatory Pathways.April 11, 20240 min read
Read more